Paragard Lawsuits
Thousands of lawsuits claim the Paragard IUD has a design flaw that causes it to break during removal, leading to severe complications and surgeries. Bellwether trials of Paragard lawsuits are set for 2025, with outcomes potentially shaping future settlements and verdicts.
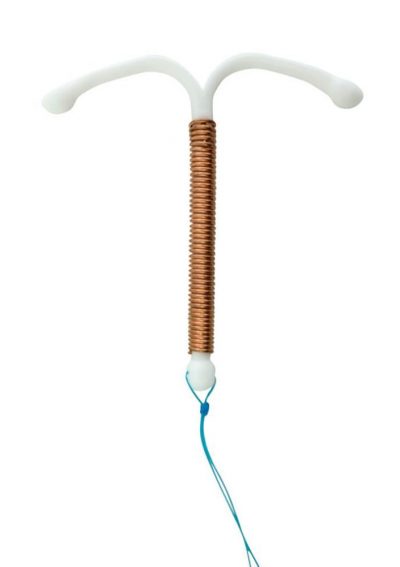
Latest Updates in Paragard Lawsuits
As of May 2026, there were 3,984 pending cases in multidistrict litigation (MDL) against Paragard. An MDL is when several similar cases are consolidated under one judge to streamline the legal process. MDL 2974 is before Judge Leigh Martin May in the Northern District of Georgia.
The plaintiffs seek compensation for injuries sustained from Paragard’s copper intrauterine device. The suits allege that manufacturers failed to warn users of the risks of a design flaw that resulted in serious complications. These complications include perforation, product migration, scarring, and, in some cases, require surgery.
Status of Paragard Lawsuits
The Paragard MDL trials have been repeatedly delayed. The first trial is currently scheduled for Jan. 2026.
-
February 2026:
Teva has prevailed in the first Paragard bellwether trial, which is an unfortunate development for plaintiffs. Two more bellwether trials remain on the schedule.
-
May 2025:
The judge overseeing the Paragard lawsuits has selected the case that will serve as the first bellwether trial. Both sides submitted options, with the judge deciding to pick a case chosen by the defendants. The first trial is set for January 2026.
-
February 2025:
Claims from several states have been dismissed due to the statute of limitations. With this ruling, the court determined that the time limit to file a claim starts when the Paragard device first causes injury inside the plaintiffs’ bodies, not when the plaintiff first discovers the injury.
Additionally, this month, plaintiffs are requesting the company to witness depositions, which are interviews with company witnesses, such as employees and representatives. These interviews occur outside of court, and the testimony may be used in trial. Witness depositions are part of the discovery process to gather information for next year's bellwether trial. There are 2,937 Paragard IUD lawsuits filed as part of the MDL. -
January 2025:
The Paragard MDL continued to grow through the end of the year, adding 38 new cases in November and another 20 lawsuits filed in December 2024. At the end of January, there were 2,882 actions now pending.
-
October 2024:
The bellwether trial dates have been rescheduled, with two trials now set for Dec. 1, 2025, and Feb. 2, 2026.
-
August 13, 2024:
Teva and the other defendants have filed a motion to dismiss numerous cases based on the statute of limitations or the statute of repose. While a statute of limitations limits the timeframe a plaintiff can file their claim, it typically has some flexibility. The statute of repose, however, has a hard deadline and may result in many cases being dismissed.
-
November 2023:
Plaintiffs were required to submit their Plaintiff Fact Sheet.
-
September 2023:
The court announced the first bellwether trial date for Oct. 28, 2024.
-
June 2023:
Bellwether cases, including depositions, were discussed in the discovery process. By the end of June, there were over 2,000 pending claims against Paragard.
-
February 2023:
Judge May outlined the bellwether selection process. The first MDL lawsuits will go to trial in 2024 unless parties settle first.
-
January 2023:
The court appointed Judge M. Gino Brogdon Jr. (retired) as a settlement mediator.
Because these lawsuits are part of an MDL, claimants keep their status as plaintiffs when the case goes before the courts or to mediation. This differs from class-action lawsuits, where one or more representative plaintiffs file a lawsuit on behalf of injured parties. MDLs only exist in the federal justice system.
Why Are People Filing Paragard IUD Lawsuits?
The Paragard IUD lawsuits all focus on an alleged design flaw that led to the IUD device breaking during removal and leaving part of the device lodged in the uterus. As a result, users claim to have endured several debilitating effects from the IUD, in some instances requiring surgery.
- Chronic pain
- Infection
- Infertility
- IUD pieces breaking and getting lodged in organs
- IUD pieces that cannot be removed from the body
- IUD pieces left in the body, causing infections and allergic reactions
- IUDs shifting position
- Need for surgical intervention such as laparoscopy or laparotomy and hysterectomy
- Perforation of the cervix or uterus
In addition to these risks of injury, research indicates people who use copper IUDs may experience an increased risk of side effects, including bacterial vaginosis and irregular menstrual bleeding. Medical researchers investigating IUD side effects have recommended improved protocols when removing Paragard IUDs.
Claims Against Manufacturers
Previous lawsuits raised serious questions about the safety of the Paragard IUD. The MDL lawsuits claim that:
- CooperSurgical and Teva Pharmaceuticals are negligent because they presented their device as safe.
- Paragard has a manufacturing defect.
- Paragard IUD has a defective design.
- Paragard’s label doesn’t clearly warn about the risks of breakage during removal.
In 2021, the U.S. Food and Drug Administration (FDA) registered over 3,000 instances of Paragard devices breaking, with 2,000 of those cases listed as severe. As of November 2024, ABC News reported over 7,000 reports of Paragard breakage.
However, Paragard’s owner, CooperSurgical, and the original manufacturer, Teva Pharmaceuticals, continue to make claims about the device’s safety and benefits.
Has There Been a Paragard IUD Recall?
The Paragard IUD was recalled in 2014 because some devices were not completely sterile. No injuries were reported as a result of this recall.
In 2019, the FDA issued the company a warning letter for false or misleading information in Paragard television ads. In 2021, the device was placed in the FDA’s Adverse Event Reporting System.
Despite the growing number of lawsuits and reported adverse events related to the Paragard IUD, neither the FDA nor the current manufacturer, CooperSurgical, have taken steps to recall the device for breakage as of May 2026.
Who Qualifies to File a Paragard Lawsuit?
If your Paragard IUD broke upon removal and you experienced severe injury or Paragard side effects, you may be eligible for compensation.
Compensation can be for injuries that cause physical, financial and emotional hardship. It can also help pay for medical treatments, replace lost wages and ease pain or suffering.
It is important to speak with a lawyer to review your options and the specifics of your case.
Case Study: Jennifer Lewis v. Teva Pharmaceuticals
Jennifer Lewis, a Calhoun County, Michigan resident, sued Teva Pharmaceuticals and its subsidiaries, as well as The Cooper Companies and CooperSurgical, over injuries she suffered after using a Paragard IUD.
Lawsuit Claims
In her complaint, Lewis claims that the Paragard IUD broke when it was being taken out in 2021, leading to internal injuries. She argues that the companies did not adequately warn users about this risk, provided misleading safety information and created a faulty product.
The main issues include negligence, strict liability and breach of warranty.
Legal Proceedings
The lawsuit was filed in September 2022 and is now part of the Paragard MDL. Lewis is seeking compensation for her injuries and punitive damages to hold the companies accountable.
Paragard Lawsuit Settlement Amounts and Verdicts
As of May 2026, there have been no trials or settlements in the Paragard MDL.
If an out-of-court settlement isn’t reached, the first trial cases will begin in early 2026.
The results of these first trial cases, called bellwether trials, can indicate the court’s eventual verdict and possible settlement amounts.
It is important to remember that a verdict is the decision in a court proceeding. A judge or a jury can deliver it, and it can include damages. A settlement is an agreement reached out of court between the two sides. The court approves it, and it can also include damages.
Plaintiffs may agree to settlements before a trial begins or once it is underway. Monetary settlements for damages can vary significantly based on the details of an individual case. Settlement negotiations usually start after the first test cases go to trial and the court’s position has been clarified.
In other IUD lawsuits, people sued Bayer for claims that its Mirena IUD moved around in the body and punctured or injured organs. Bayer offered a $12.2 million settlement to settle 4,600 lawsuits.
Editor Lindsay Donaldson contributed to this article.
19 Cited Research Articles
Consumernotice.org adheres to the highest ethical standards for content production and references only credible sources of information, including government reports, interviews with experts, highly regarded nonprofit organizations, peer-reviewed journals, court records and academic organizations. You can learn more about our dedication to relevance, accuracy and transparency by reading our editorial policy.
- United States Judicial Panel on Multidistrict Litigation. (2026, May 1). MDL Statistics Report - Distribution of Pending MDL Dockets by Actions Pending. Retrieved from https://www.jpml.uscourts.gov/sites/jpml/files/Pending_MDL_Dockets_By_Actions_Pending-May-1-2026.pdf
- U.S. District Court for the Northern District of Georgia. (2025, April 29). Order. Retrieved from https://ecf.gand.uscourts.gov/doc1/055117223060
- United States Judicial Panel on Multidistrict Litigation. (2024, December 2). MDL Statistics Report. Retrieved from https://www.jpml.uscourts.gov/sites/jpml/files/Pending_MDL_Dockets_By_MDL_Number-January-2-2025.pdf
- Moreschi, A., et al. (2024, November 14). FDA Launched Safety Review Into Broken IUDs After Spotlight Inquiry, but Questions Remain. Retrieved from https://abc3340.com/news/spotlight-on-america/spotlight-inquiry-led-to-fda-safety-review-of-paragard-breakage-but-questions-remain
- United States District Court, Northern District of Georgia, Atlanta Division. (2024, October 29). Minute Sheet for Proceedings Held in Chambers on 10/29/2024. Retrieved from https://ecf.gand.uscourts.gov/doc1/055116737749
- U.S. District Court for the Northern District of Georgia, Atlanta Division. (2024, January 2). Case Management Order and Second Amended Scheduling Order. Retrieved from https://www.aboutlawsuits.com/wp-content/uploads/2024-01-02-Order.pdf
- Shandruk, E. & Cellucci, L. (2023, May 20). MDL vs. Class Action: Place, Plaintiffs and Procedure. Retrieved from https://www.americanbar.org/groups/young_lawyers/resources/tyl/practice-areas/mdl-vs-class-action-place-plaintiffs-and-procedure/
- Daniel, A. et al. (2023, April). An Integrative Review of the Relationship Between Intrauterine Devices and Bacterial Vaginosis. Retrieved from https://www.sciencedirect.com/science/article/abs/pii/S1751485123000399?via%3Dihub
- Baker, C. & Creinin, M. (2022, November). Long-Acting Reversible Contraception. Retrieved from https://journals.lww.com/greenjournal/Abstract/2022/11000/Long_Acting_Reversible_Contraception.24.aspx
- Christelle, K., et al, (2022, August 26). Interventions to Prevent or Treat Heavy Menstrual Bleeding or Pain Associated With Intrauterine-Device Use. Retrieved from https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD006034.pub3/full
- Lanzola, E. & Ketvertis, K. (2022, July 4). Intrauterine Device. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK557403/
- U.S. Food and Drug Administration. (2022, April 6). October - December 2021 | Potential Signals of Serious Risks/New Safety Information Identified by the FDA Adverse Event Reporting System (FAERS). Retrieved from https://www.fda.gov/drugs/questions-and-answers-fdas-adverse-event-reporting-system-faers/october-december-2021-potential-signals-serious-risksnew-safety-information-identified-fda-adverse
- Valderrama, S. (2022, February 12). I-Team Investigates After Thousands of Women File Lawsuit Against IUD Maker. Retrieved from https://weartv.com/news/local/i-team-investigates-after-thousands-of-women-file-lawsuit-against-iud-maker
- Sterman, J., et al. (2021, August 2). As Reports of IUD Breakage Piled Up, Maker Changed Label but Many Women Still Unaware. Retrieved from https://katv.com/news/spotlight-on-america/as-reports-of-iud-breakage-piled-up-maker-changed-label-but-many-women-still-unaware
- Sarver, J., et al. (2021, January 23). Fractured Copper Intrauterine Device (IUD) Retained in the Uterine Wall Leading to Hysterectomy: A Case Report. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7851793/
- United States Judicial Panel on Multidistrict Litigation. (2020, December 16). In re: Paragard IUD Products Liability Litigation. Retrieved from https://www.jpml.uscourts.gov/sites/jpml/files/MDL-2974-Transfer%20Order-12-20.pdf
- Dubovis, M. & Rizk, N. (2020, April 24). Retained Copper Fragments Following Removal of a Copper Intrauterine Device: Two Case Reports. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7180360/
- U.S. Department of Health and Human Services. (2017, February 15). Human Drug Product Recalls Pending Classification. Retrieved from https://wayback.archive-it.org/7993/20170403231827/https://www.fda.gov/Safety/Recalls/EnforcementReports/ucm310739.htm
- CooperSurgical. (n.d.). Paragard (Intrauterine Copper Contraceptive) The Only One for Almost Everyone. Retrieved from https://www.coopersurgical.com/our-brands/paragard/
Calling this number connects you with a Consumer Notice, LLC representative. We will direct you to one of our trusted legal partners for a free case review.
Consumer Notice, LLC's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.
844-891-3125
