Exactech Lawsuit
Lawyers are still taking cases, but Exactech's October 2024 bankruptcy filing has paused ongoing court actions tied to defective joint replacements, affecting trial schedules. With over 143,000 recalled implants in question, patients are pursuing compensation for injuries like device failure and revision surgeries.
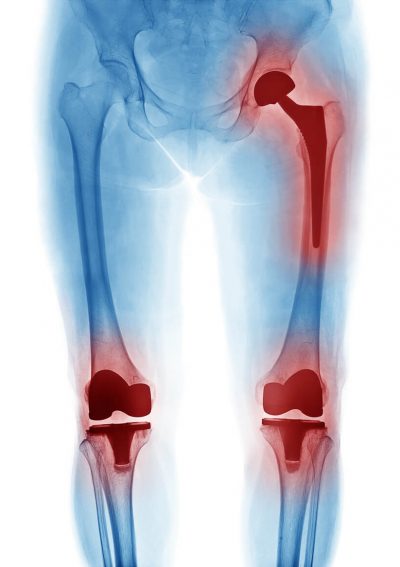
Latest Exactech Lawsuit Updates
In October 2024, Exactech filed for Chapter 11 bankruptcy in response to mounting lawsuits against the company. The bankruptcy filing has paused ongoing litigation against the company.
As of April 2026, there were 1,838 Exactech lawsuits pending in multidistrict litigation before the U.S. District Court, Eastern District of New York. So far, no trials or approved settlements in MDL 3044 exist.
The court modified the bellwether trial schedule earlier in October 2024:
- The Tarloff trial was set to start on Sept. 29, 2025.
- The Larson trial was scheduled to begin on Nov. 10, 2025.
However, Exactech’s bankruptcy filing may further affect these trial dates.
-
November 2024
Case is stayed, meaning temporarily paused, because of bankruptcy proceedings.
-
October 2024
Exactech has filed for bankruptcy in response to the mounting litigation. The court has paused pending lawsuits against Exactech due to the bankruptcy filing.
-
August 2024
The start of the first two bellwether trials in the MDL has been moderately delayed following ongoing discovery disputes. Gayle Tarloff’s trial is now set to begin on July 6, 2025, with Geraldine Larson’s trial moving to Sep. 29, 2025. The trials had previously been set for June and August of that year.
-
April 2024
Courts set the initial bellwether trial date for June 2025 to try plaintiff Gayle Tarloff’s case.
-
October 2023
On Oct. 20, the judge lifted the stay on a pending transfer order from Oct. 7, and 20 more cases were transferred into MDL 3044 and assigned to U.S. District Judge Nicholas G. Garaufis.
-
March 2023
According to Magistrate Judge Marcia M. Henry's order, all MDL plaintiffs must complete Plaintiff Facts Sheets before June 6, 2023.
-
January 2023
Magistrate Judge Henry signed a protective order that had been agreed upon and signed by both the plaintiffs' and defendants' counsel. The order spelled out how to handle confidential documents that will be part of discovery in the litigation.
-
October 2022
The Panel on Multidistrict Litigation approved the request to consolidate cases from around the country and transfer them to the Eastern District of New York, with Judge Garaufis overseeing coordinated proceedings.
The plaintiffs in the Exactech lawsuit seek compensation for injuries related to joint replacement devices. In March 2023, the FDA reminded medical providers and the public about the defective packaging for many of Exactech’s joint replacement devices manufactured between 2004 and August 2021. Defective packaging could cause the devices to degrade early, leading to revision surgery.
What Are Exactech Lawsuits?
Exactech lawsuits are product liability claims asking that Exactech compensate those injured by the company’s defective knee, ankle and hip implants. In 2021 and 2022, Exactech recalled its Ultra-High Molecular Weight Polyethylene Knee and Ankle Polyethylene Inserts manufactured between 2004 and August 2021 because product packaging was not up to specifications. Truliant knee, Optetrak knee and Vantage ankle devices used these inserts.
“The defective bags were missing one of the oxygen barrier layers that protect devices from oxidation, a chemical reaction with oxygen that can degrade plastics over time. Oxidation can lead to accelerated device wear/failure, and component cracking or fracture, all leading to corrective revision surgery,” according to the U.S. Food and Drug Administration’s March 23, 2023, safety communication.
In June 2021, Exactech recalled some GXL liners used in MCS, Acumatch and Novation hip implants because of premature and excessive wear. However, the company didn’t provide a reason for the wear problems. In August 2022, the company expanded its original recall to include all devices with plastic components packaged in defective bags.
By March 2024, Exactech voluntarily recalled its Equinoxe Shoulder System implants for the same packaging defects.
Exactech Lawsuit Settlements
Some lawyers estimate a potential Exactech lawsuit settlement to be between $100,000 and $300,000. This projection is based on knee replacement settlements from past lawsuits. A claimant could receive a smaller or larger settlement depending on the injuries claimed.
So far, there have been no court-approved settlements for Exactech lawsuits. Typically, bellwether trials determine potential settlement amounts. The first bellwether trial is scheduled for June 2025.
Why Are People Filing Exactech Replacement Lawsuits?
People are filing Exactech lawsuits to get compensation for damages incurred because of potentially defective Exactech hip, knee or ankle implants. They claim Exactech knew these products were faulty, but the company still sold them, making it guilty of negligence, among other claims.
- Breach of warranty
- Failure to warn
- Fraud
- Negligence
- Strict product liability
According to Exactech’s April 2022 Urgent Medical Device Correction letter, at least 143,484 potentially defective inserts were implanted in the U.S. These devices could have put tens of thousands of Americans at risk of early failure after Exactech knee or ankle replacements.
Injuries Claimed in Exactech Lawsuits
Those who received a recalled Exactech implant could face many painful complications. When devices fail early, symptoms may include pain, bone loss and problems with mobility. Sometimes, the only way to correct these issues is through revision surgery.
- Bone loss
- Component fracture
- Device failure
- Difficulty walking
- Dislocations
- Early and excessive device wear
- Inability to bear weight
- Instability
- New or worsening pain
- Noises such as clicking, grinding or popping
- Revision surgery
- Swelling
“Recipients of the Optetrak knee implants have required painful revision surgeries well before the estimated life expectancy of the devices, and at a much higher rate than should reasonably be expected for devices of this kind,” according to one Exactech lawsuit filed by Joseph D. Sexton and his wife, Corrine.
Case Study: Michael Giordano’s Injuries From a Faulty Knee Replacement
Michael Giordano of Woodbury, Conn., had surgery to replace his left knee on Jan. 20, 2015. The operation was performed at a hospital in New York City known for its expertise in joint surgeries. During this operation, Giordano received an Exactech knee implant that had previously been recalled due to safety concerns.
- Complications After Surgery
- In 2020, Giordano faced severe problems with the implant. It was wearing out faster than expected, leading to another surgery to replace it. The new implant used was also from the same recalled group. Even after this second surgery, Giordano continues to experience severe pain, swelling and difficulty walking, according to his Exactech lawsuit complaint. He struggles with balance and has ongoing issues with his gait, resulting in bone loss and damage to the soft tissues around his knee.
- Long-Term Problems Continue
- These health issues have greatly affected Giordano's daily life, requiring consistent medical attention and leaving him in need of yet another surgery in the future. According to Giordano's attorney, his condition has caused him emotional distress and financial strain, as he has faced a loss of income due to his inability to work. Court documents claim the problems caused by these faulty knee replacement devices have not only harmed Giordano's physical health but have also impacted his independence and overall well-being, resulting in long-term financial and emotional challenges.
Who Can File an Exactech Lawsuit?
If you received a recalled Exactech knee, hip or ankle implant and had surgery or your health care provider advised you to have surgery, you may qualify to file an Exactech lawsuit. Check your medical records or speak with your orthopedic surgeon to verify the implant you received.
If you are unsure what brand of implant you have, you can seek the assistance of a product liability lawyer. Lawsuits include devices manufactured between 2004 and August 2021.
- Acumatch Hip with Connexion GXL liner
- MCS Hip with Connexion GXL liner
- Novation Hip with Connexion GXL liner
- Optetrak Knee: All-polyethylene CR Tibial Components, All-polyethylene PS Tibial Components, CR Tibial Inserts, CR Slope Tibial Inserts, PS Tibial Inserts, HI-FLEX PS Tibial Inserts
- Optetrak Logic Knee: CR Tibial Inserts, CR Slope Tibial Inserts, CRC Tibial Inserts, PS Tibial Inserts, PSC Tibial Inserts, CC Tibial Inserts
- Truliant Knee: CR Tibial Inserts, CR Slope Tibial Inserts, CRC Tibial Inserts, PS Tibial Inserts, PSC Tibial Inserts
- Vantage Ankle: Fixed-Bearing Liner Component
- Limited mobility
- Loosening because of osteolysis
- Pain
- Radiographic evidence of osteolysis
- Stiffness
Exactech advised doctors to contact patients who were implanted with recalled devices. But if you haven’t received a call from your doctor, contact their office to schedule a follow-up appointment.
What Should You Do if an Exactech Replacement Injured You?
If you suffered complications or injuries from an Exactech replacement, schedule an evaluation with your doctor. If you received a recalled implant, contacting your doctor, regardless of any current problems, is a good idea. Some people may not have symptoms even if the device is degrading prematurely.
Your doctor may schedule surgery if the implant is causing problems or has failed. However, the FDA doesn’t recommend surgery for people who aren’t having issues with their implants.
Exactech offers direct compensation to those affected by recalled implants, but signing any paperwork with Exactech may prevent you from filing a lawsuit against them. Before contacting Exactech, speak to a lawyer about your legal rights. Lawyers typically offer free case evaluations without obligation for people injured by Exactech knee, hip or ankle implants.
Editor Lindsay Donaldson contributed to this article.
15 Cited Research Articles
Consumernotice.org adheres to the highest ethical standards for content production and references only credible sources of information, including government reports, interviews with experts, highly regarded nonprofit organizations, peer-reviewed journals, court records and academic organizations. You can learn more about our dedication to relevance, accuracy and transparency by reading our editorial policy.
- United States Judicial Panel on Multidistrict Litigation. (2026, April 1). MDL Statistics Report - Distribution of Pending MDL Dockets by Actions Pending. Retrieved from https://www.jpml.uscourts.gov/sites/jpml/files/Pending_MDL_Dockets_By_Actions_Pending-April-1-2026.pdf
- Randles, J. (2024, October 31). TPG’s Exactech Goes Bankrupt After Lawsuits Over Recalled Implant Devices. Retrieved from https://www.insurancejournal.com/news/southeast/2024/10/31/799287.htm
- Exactech. (2024, October 29). Exactech Reaches Agreement With Investor Group to Build Foundation for Future Growth. Retrieved from https://www.prnewswire.com/news-releases/exactech-reaches-agreement-with-investor-group-to-build-foundation-for-future-growth-302289670.html
- U.S. District Court for the Eastern District of New York. (2024, August 16). Re: MDL No. 3044, in re: Exactech Polyethylene Orthopedic Products Liability Litigation, 1:22-md-03044-NGG-MMH – Pretrial Schedules for Tarloff and Larson Bellwethers. Retrieved from https://ecf.nyed.uscourts.gov/doc1/123121604977
- U.S. Food and Drug Administration. (2024, April 19). Risks With Exactech Equinoxe Shoulder System With Defective Packaging. Retrieved from https://www.fda.gov/medical-devices/safety-communications/risks-exactech-equinoxe-shoulder-system-defective-packaging-fda-safety-communication
- U.S. District Court for the District of Connecticut. (2023, April 11). Giordano v. Exactech, Inc., et Al.; Complaint & Demand for Jury Trial, Case 3:23-cv-00457-JCH. Retrieved from http://www.pacer.gov
- U.S. Food and Drug Administration. (2023, March 23). Risks With Exactech Joint Replacement Devices With Defective Packaging – FDA Safety Communication. Retrieved from https://www.fda.gov/medical-devices/safety-communications/risks-exactech-joint-replacement-devices-defective-packaging-fda-safety-communication
- United States Judicial Panel on Multidistrict Litigation. (2023, March). Plaintiff Facts Sheets Order. Retrieved from https://ecf.jpml.uscourts.gov/cgi-bin/show_temp.pl?file=1140914-0--41477.pdf&type=application/pdf
- Sexton v. Exactech Inc. (2022, November 11). Complaint and Jury Demand. Retrieved from https://www.aboutlawsuits.com/wp-content/uploads/2022-11-11-exactech-sexton-complaint.pdf
- United States Judicial Panel on Multidistrict Litigation. (2022, October 7). MDL Transfer Order. Retrieved from https://www.jpml.uscourts.gov/sites/jpml/files/MDL-3044-Initial-Transfer-Order-9-22.pdf
- Exactech. (2022, April 7). Exacttech DHCP Letter. Retrieved from https://www.exac.com/wp-content/uploads/2022/04/Exactech-DHCP-letter.4.6.2022.pdf
- Exactech. (2022, February 7). Exactech DHCP Letter. Retrieved from https://www.exac.com/product/exactech-dhcp-letter/
- Exactech. (2022, February 7). Frequently Asked Questions: Knee and Ankle Recall. Retrieved from https://www.exac.com/wp-content/uploads/2022/02/Exactech-Knee-and-Ankle-FAQ.02.07.2022_Letterhead.pdf
- Exactech. (2022, February 7). US Recall Information, Knee/ankle. Retrieved from https://www.exac.com/medical-professionals/recall-information/
- Exactech. (2021, June 24). FAQ – GXL Liners for Novation, Acumatch and MCS Systems. Retrieved from https://www.exac.com/wp-content/uploads/2021/06/Frequently-Asked-Questions-FAQs-for-US-Agents-Surgeons_FINAL_GXL.pdf
Calling this number connects you with a Consumer Notice, LLC representative. We will direct you to one of our trusted legal partners for a free case review.
Consumer Notice, LLC's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.
844-420-1914



