Drugs & Devices
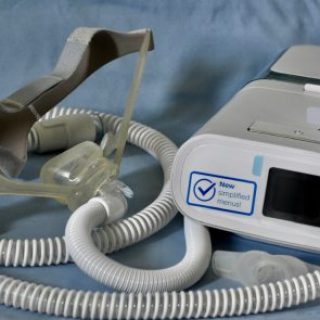
Philips Respironics Halts U.S. CPAP Sales
Philips Respironics ceases new U.S. CPAP, BiPAP sales. Discover more about the FDA settlement and ongoing safety measures. Read now for details.